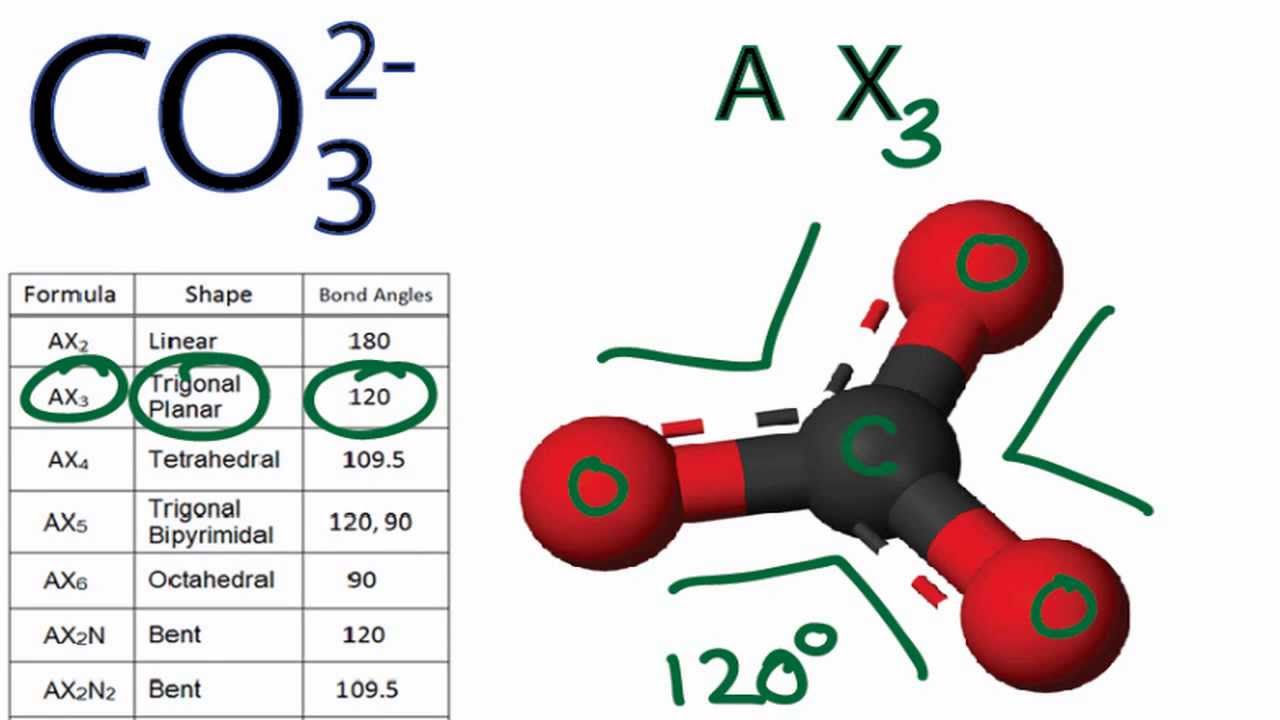
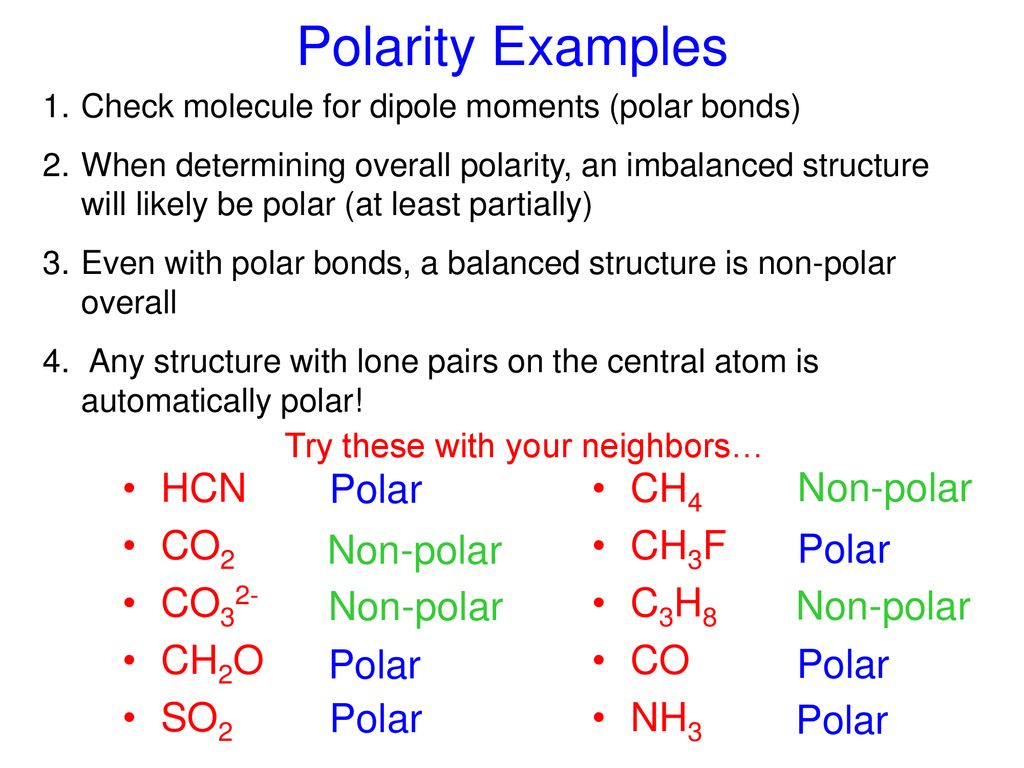
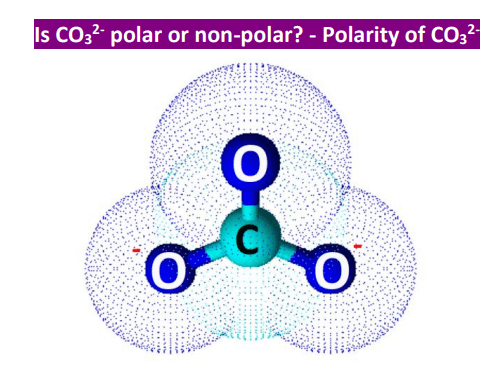
Can a molecule (like CO3 2-) be nonpolar even if it has two single bonds and one double bonds? - Quora
amongst NO3^ 1, AsO3^ 3,CO3^ 2, ClO3^ 1, SO3^ 2 and BO3^ 3, the non polar species are: (1) NO3^ 1,CO3^ 2, BO3^ 3 (2) AsO3^ 3, ClO3^ 1, SO3^ 2 (3) CO3^ 2, SO3^ 2, AsO3 3 (4) ClO3^ 1, SO3^ 2, NO3^ 1

Determine if each compound or ion below has a dipole moment. (a) Carbonate ion (CO_3 ^{2-}) | Homework.Study.com



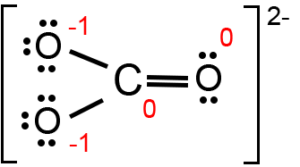
![SOLVED: Which of the following molecules is polar? [SO3]2 - [SbF6] - [PF4]+ [SiO4]4 - [CO3]2 - SOLVED: Which of the following molecules is polar? [SO3]2 - [SbF6] - [PF4]+ [SiO4]4 - [CO3]2 -](https://cdn.numerade.com/ask_images/726bca042e434fde93424aecaa925380.jpg)